 
When beryllium forms a linear molecule such as beryllium chloride, it is not the sp hybrids themselves that form the two bonds but rather an overlap between each of these orbitals and some orbital on each other atom. Since both correspond to the same physical reality, we can use whichever approach suits us best. By contrast it is much easier to explain the periodic table by using s and p orbitals rather than hybrids. It is much easier to think of two Be-Cl bonds separated by 180° in terms of sp hybrids than in terms of separate s and p orbitals. Principle energy levels are color coded, while sublevels are grouped together and each circle represents an orbital capable of holding two electrons. Different groups are shown in different colors. In other words, it does not matter to the molecule whether we think of the total electron cloud as being formed from s and p orbitals or from sp hybrids, but it does matter to us. 5: Electrons are added to atomic orbitals in order from low energy (bottom of the graph) to high (top of the graph) according to the Aufbau principle. How Is the Periodic Table Organized Diagram of The Periodic Table of Elements. Labeling one electron cloud as s and another as p is an aid to our thinking, just as color-coding one sp-hybrid electron density red and the other gray is, but it is the overall electron density which determines the experimentally observable molecular geometry. In actual fact all electrons are identical-we cannot distinguish one from another experimentally. To see the elongated shape of (x, y, z)2 functions that show. Each picture is domain coloring of a (x, y, z) function which depends on the coordinates of one electron. The two colors show the phase or sign of the wave function in each region. Rather, we are looking at the same electron density, but we have color coded it to emphasize its concentration along the x axis. The shapes of the first five atomic orbitals are: 1s, 2s, 2p x, 2p y, and 2p z. The second shell has 2 subshells: 1 s-orbital and 3 p-orbitals. This means that the first shell can hold 2 electrons. 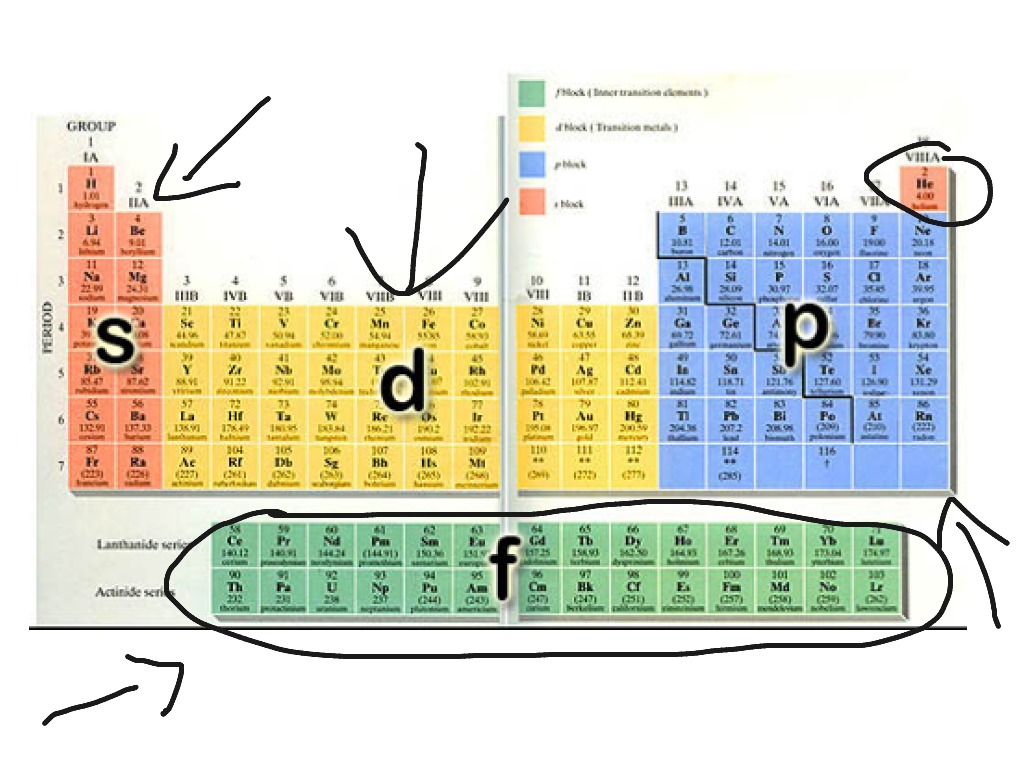
The first shell (of all atoms) has 1 subshell of s-orbitals containing 1 s orbital. We have not created something new with the two sp hybrids. Every subshell has a of orbits s/p/d/f that can each hold 2 electrons each (one has the opposite spin of the other). That is, the overall electron density (due to the four electrons occupying two orbitals) is exactly the same in both cases. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
